Lodhia, V., Hautus, M., Johnson, B. W., & Brock, J. (in press). Atypical brain responses to auditory spatial cues in adults with autism spectrum disorder. European Journal of Neuroscience.
The auditory processing atypicalities experienced by many individuals on the autism spectrum disorder might be understood in terms of difficulties parsing the sound energy arriving at the ears into discrete auditory “objects.” Here we asked whether autistic adults are able to make use of two important spatial cues to auditory object formation – the relative timing and amplitude of sound energy at the left and right ears. Using electroencephalography, we measured the brain responses of 15 autistic adults and 15 age-and verbal-IQ-matched control participants as they listened to dichotic pitch stimuli – white noise stimuli in which interaural timing or amplitude differences applied to a narrow frequency band of noise typically lead to the perception of a pitch sound that is spatially segregated from the noise. Responses were contrasted with those to stimuli in which timing and amplitude cues were removed. Consistent with our previous studies, autistic adults failed to show a significant Object Related Negativity (ORN) for timing-based pitch, although their ORN was not significantly smaller than that of the control group. Autistic participants did show an ORN to amplitude cues, indicating that they do not experience a general impairment in auditory object formation. However, their P400 response – thought to indicate the later attention-dependent aspects of auditory object formation – was missing. These findings provide further evidence of atypical auditory object processing in autism with potential implications for understanding the perceptual and communication difficulties associated with the condition.
Introduction
Autism spectrum disorder (ASD) is characterised by impaired social interaction, verbal and non-verbal communication co-occurring with restricted behaviours and interests (American Psychiatric Association, 1994; American Psychiatric Association, 2013). However, sensory symptoms and deficits are more common in children with ASD compared to children with other developmental disorders (Rogers & Ozonoff, 2005) and have recently been added to diagnostic criteria. Atypical auditory sensitivity is particularly common, with numerous accounts of distressing hyper-reactivity to noise (Grandin & Scariano, 1986; Landon et al., 2016; Rosenhall et al., 1999; Talay-Ongan, & Wood, 2000). Of particular note, several studies have reported that individuals with ASD have difficulty extracting relevant auditory information (i.e., speech or targets) in the presence of competing background noise (Alcantara et al., 2012; Alcantara et al., 2004; Boatman et al, 2001; Groen, et al 2009; Teder-Salejarvi et al., 2005). Observations such as these suggest that autistic individuals may have difficulty with “auditory scene analysis,” the process by which the auditory information entering the two ears is parsed into discrete auditory ‘objects’ corresponding to different sound sources in the environment (Bregman, 1990).
In the current study, we used behavioural and electrophysiological methods to investigate autistic adults’ use of spatial cues to parse acoustic stimuli into distinct objects. The human auditory system determines the spatial origin of a sound using two cues – the relative loudness in each ear and the relative timing (i.e., phase) of the sound in each ear. For example; a sound to the left of midline is louder and arrives slightly earlier in the left ear than the right. Studies in humans (Pratt, et al., 1997) and other animals (Smith et al., 1993; Yin & Kuwada, 1984) suggest that these cues are processed independently in the brainstem and may only be combined in the later stages of cortical processing (Johnson & Hautus, 2010).
We presented participants with dichotic pitch stimuli (Johnson et al., 2003; Johnson & Hautus, 2005; Clapp et al., 2007) – stereo broadband noise in which a narrow frequency band is shifted in time or amplitude between the left and right channels. These inter-aural timing (ITD) or amplitude (IAD) differences typically result in the perception of a pitch at that frequency in a different location to the rest of the noise (Bilsen, 1976; Cramer & Huggins, 1958, Dougherty et al., 1998). Previous electroencephalography (EEG) studies contrasting the response to dichotic pitch stimuli and control stimuli in which no pitch sound is perceived, have identified two components of the difference waveform (Alain et al., 2001; Clapp et al., 2007; Hautus & Johnson, 2005; Johnson et al., 2007). The “object related negativity” (ORN) at around 250 ms occurs during both passive and active listening, suggesting that it reflects a pre-attentive stage of sound segregation. When participants are attending to the stimuli, the ORN is followed at around 400 ms by a positivity referred to as the P400, thought to reflect top-down processes involved with perceptual decision making (Alain et al, 2001; Johnson et al., 2007; Hautus et al., 2009).
In a recent study using ITD-cued dichotic pitch (Lodhia et al., 2014), we found that autistic adults performed worse than control participants when required to indicate the presence or absence of the tone. They also showed a significantly weaker ORN than control participants and failed to show a significant P400. We found broadly consistent results in a second study in which we measured brain responses during passive listening using magnetoencephalography. Typically developing adults showed a differential response to dichotic pitch and control stimuli, but this effect was absent in a group of age-matched autistic children (Brock et al., 2013). Together, these findings suggest that autistic individuals have difficulty using ITDs to segregate the auditory environment into distinct objects. What remains unclear, however, is whether this difficulty is specific to ITD cues or whether it reflects a more general impairment in auditory object formation.
Thus, in the current study, we contrasted behavioural and brain responses to IAD as well as ITD dichotic pitch stimuli. Based on our previous studies, we expected that for ITD stimuli, the ORN (and subsequent P400) would be absent in autistic adults. If this reflects a general problem with auditory scene analysis, then we should also obtain a reduced ORN for IAD stimuli. However, if there is specifically a problem with ITDs then the ORN should be present for IAD stimuli. This second scenario also gave us an opportunity to investigate the P400. If the P400 is absent or reduced following an intact ORN, this would indicate an impairment in the later attention-dependent component of auditory object formation.
Materials and methods
Ethics statement
The study conforms with the World Medical Association Declaration of Helsinki. All participants gave their informed written consent and all procedures were approved by The University of Auckland Human Participants Ethics Committee (2009/537).
Participants
The ASD participants were 15 adults (13 males, 2 females) recruited via adverts posted at Autism NZ, Altogether Autism, Autism House, Centre for Brain Research, and the University of Auckland. Each participant in the ASD group was given a clinical diagnosis of autistic disorder (N = 3) or Asperger’s Disorder (N = 12) according to DSM-IV criteria. As such, they would all qualify for a DSM-5 diagnosis of Autism Spectrum Disorder. Diagnoses were made by a clinical psychologist or paediatrician. In addition to the clinical diagnoses, all ASD participants met the cut-off for ASD on the Social Communication Questionnaire (SCQ – Lifetime scale ≥15), which was completed by a parent or guardian at the first study meeting (Berument et al., 1999).
The TD group were 15 adults (13 males, 2 females) with no personal or family history of neurological or psychiatric disorders, who were recruited via the University of Auckland Psychology Research participation website and posters placed around the University of Auckland city campus. They were matched to the ASD group on gender, age (± 2 years), and handedness as determined by the Edinburgh Handedness Inventory (Oldfield, 1971).
Exclusion criteria for both groups included a co-morbid axis 1 disorder or relevant axis 3 diagnosis, hearing deficits or pharmacological treatment. Further inclusion criteria were (1) normal auditory acuity – hearing thresholds ≤ 25 dB HL, as assessed by an audiometer (Amplitude T-Series, Otovation, LLC USA) for the standard range of 250-8000 Hz; (2) a full-scale mental ability score whose lower confidence bound was ≥ 80 (Wechsler Abbreviated Scales of Intelligence, 1999); and (3) passing a pre-screening assessment (see below) demonstrating an ability to detect ITD dichotic pitch. Table 1 summarizes the demographic and behavioural test data for both groups.
| ASD (N=15)
M (SD) |
TD (N=15)
M (SD) |
t (28) | p | |
| Age | 25.80 (6.81) | 27.07 (5.80) | 0.55 | 0.59 |
| Handedness | 61.53 (67.81) | 60.00(82.81) | -0.06 | 0.96 |
| Verbal IQ | 123.40 (13.44) | 130.53 (9.09) | 1.70 | 0.10 |
| Performance IQ | 116.93 (13.95) | 119.40 (16.79) | 0.44 | 0.67 |
| Combined IQ | 122.93 (12.83) | 129.07 (7.40) | 1.60 | 0.12 |
| SCQ lifetime score | 19.67 (4.65) | – | – | – |
Table 1: Demographic and cognitive characteristics of the TD and ASD groups
Stimuli
Auditory stimuli were generated during the experiment on two-channels of a 16-bit converter (Model NIUSB-6259, National Instruments, Austin, TX). Stimulus creation began with the generation of two independent 500ms broadband Gaussian noise bursts, each 500 ms in duration at a sampling rate of 44.1 kHz, using LabView software (National Instruments, Austin, Texas, USA). One noise burst was bandpass filtered with a centre frequency of 600 Hz and a bandwidth of 20 Hz using a fourth-order Butterworth filter. The other noise burst was notch filtered using the same filter characteristics. A copy was made of both noises (bandpass and notch) for each ear.
For ITD pitch stimuli, opposing inter-aural delays of ±500 μs were applied to the bandpass- and notch-filtered noises so that the resulting combination would create a noise lateralized to one side of auditory space and a pitch to the other side of auditory space. For ITD control stimuli, both the bandpass-and the notch-filtered noise were interaurally delayed (500 μs) to the same ear.
The IAD stimuli were adapted for each participant (see matching task described below). Stimuli were created by adjusting the relative intensity of the left and right channels of the bandpass- and the notch-filtered noise stimuli whilst maintaining the summed amplitude of the left and right channels for each component at a constant level. For IAD pitch stimuli, the bandpass- and notch-filtered noises were lateralized to opposite sides. For IAD control stimuli, the bandpass- and notch-filtered noise were co-lateralized.
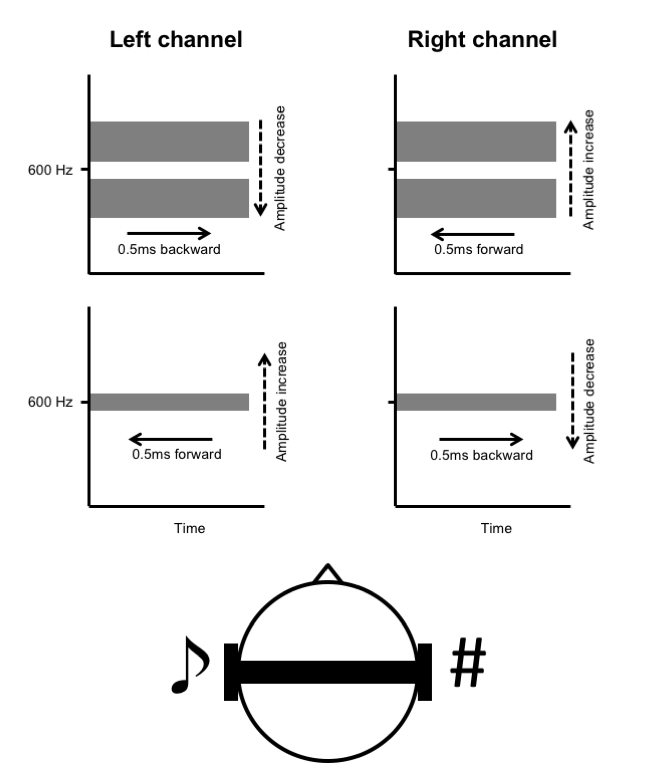
The final stage in creating each stimulus was to recombine the bandpass and notch-filtered noises for each channel. The resultant stimulus was then bandpass filtered (fourth-order Butterworth filter) with a centre frequency of 600 Hz and bandwidth of 400 Hz and windowed with a cos² function with 4 ms rise and fall times. Programmable attenuators (Model PA4, Tucker-Davis Technologies, Alachua, FL) set the binaural stimuli to yield 70 dB SPL from insert earphones at the ear (ER2, Etymotic Research Inc., Elk Grove Village, Illinois, USA). A similar method of ITD and IAD dichotic pitch construction was used in a study with TD adults (Johnson and Hautus, 2010). Figure 1 shows a schematic representation of ITD and IAD dichotic pitch stimuli used in the experiment and the resulting listener percept’s.
Procedure
Screening task
To ensure that all participants could perceive the ITD, participants completed a practice session of four 100 trial blocks with ITD stimuli. On each trial, they had to indicate via button press whether the stimulus presented contained one or two auditory objects. Feedback was provided after each trial. All participants met the criterion of 69 percent correct (approximately d′ = 1; cf. Macmillan & Creelman, 2005, p. 9) in order to proceed to the matching task.
Matching task
Prior to the EEG recording, all participants completed a matching task to ensure that the ITD and IAD stimuli were perceptually as similar as possible. On each trial, participants heard an ITD stimulus followed by an IAD stimulus that was lateralized to the same side of space by a randomly chosen amount. Participants gave a keypress response to indicate the direction the IAD stimulus should move so that it more closely matched their perception of the ITD stimulus. Pressing 1 on the keyboard moved the IAD stimulus 10% to the left. Pressing 2 moved it 10% to the right. They then heard the ITD stimulus again, followed by the new IAD stimulus. The trial continued until the participant indicated that they were satisfied with the match by pressing 3 on the keyboard.
The matching task consisted of four blocks each of 16 trials, with the average final value across the 16 trials being used to determine the IAD stimulus in the corresponding condition of the EEG behavioural task (see below). In Blocks 1 and 2, participants matched IAD control stimuli to the fixed ITD control stimuli. Stimuli were left-lateralized in Block 1 and right-lateralized in Block 2. In Blocks 3 and 4, participants matched the pitch (i.e., band-pass filtered component) of the IAD pitch stimulus to the fixed ITD pitch stimulus with the (notch-filtered) noise component of the IAD stimulus held constant at the values obtained from Blocks 1 and 2. The pitches were right-lateralized in Block 3 and left-lateralized in Block 4.
EEG behavioural task
For the EEG recording, participants heard a single stimulus and had to decide whether it contained one sound object or two by pressing the 1 or 2 key respectively. No feedback was provided and the trial timed out after 1500ms if no response was made. The interval between response / timeout and the next stimulus was drawn from a rectangular distribution between 2000 ms and 3400 ms. Participants completed 1440 trials, split into four blocks with short breaks between blocks. Each block comprised 90 ITD Pitch stimuli, 90 IAD Pitch stimuli, 90 ITD control stimuli, and 90 IAD control stimuli and took approximately 20 minutes to complete.
EEG recording
Participants were tested in an electrically shielded room (Model L3000; Belling Lee, Enfield, England). EEG data were recorded continuously at 250-Hz (0.1–100 Hz analogue bandpass) using 128-channel Ag/AgCl electrode nets (Tucker, 1993; Electrical Geodesics Inc., Eugene, Oregon, USA) and Electrical Geodesics Inc. amplifiers (200-MΩ input impedance). Electrode impedances were kept below 40 kΩ (Tucker, 1993). Common vertex (Cz) was used as a reference. During the EEG, participants were asked to fixate on a cross, presented on a computer screen, in order to minimise eye-movement artifacts.
Data and statistical analysis
EEG recordings were processed using custom in house software. The recordings were segmented into 850 ms epochs including a 100 ms pre-stimulus baseline. All ocular artifacts were corrected (Jervis et al., 1985) and trials with bad channels were dropped. Approximately 3% of trials were lost for both groups. We included all trials, regardless of whether the behavioural response was correct or incorrect (if we assume that the perceptual processes indexed by the ORN and P400 contribute to subsequent behaviour, removing error trials entails removing the effect of interest). Responses for each stimulus were then averaged together, digitally filtered with a zero-phase-shift 3-pole Butterworth filter (0.1–30 Hz; Alarcon et al., 2000), and then re-referenced to the mean.
Channel selection for statistical analysis was based on the grand mean difference waveform, derived by subtracting the control from the pitch stimulus and averaging across groups and conditions. This biases our results towards finding an overall main effect, but is independent of group and condition. We first identified two peaks corresponding to the ORN and P400 in the global field power of the grand mean difference waveform. Topographies of the ORN and P400 were similar, with a circular cluster of 15 electrodes, centred on CZ (6, 7, 13, 30,31, 37, 54, 55, 79, 80, 87, 105, 106, 112, 129) showing the greatest effect of Pitch at both time points. Waveforms plotted below were derived from the average of these channels. Next, we determined time windows for the ORN and P400 based on the full width half max of the two components. Statistical analyses were then performed on the area under the curve within these two windows (see R Markdown for full details: http://rpubs.com/JonBrock/DichoticPitch).
Results
Behavioural performance
Matching task
Figure 2a shows the average response for the different conditions of the matching task. We conducted a Group by Stimulus (Noise vs Tone) by Laterality ANOVA on the absolute value of responses. There was no effect of Group, F(1,28) = 1.03, p = .320, ges = 0.025. We also found no effects of Stimulus (Noise vs Tone), Laterality, and no interactions (Fs < 1.1, ps > 0.3). This indicates that the two groups had comparable sensitivity to ITD relative to IAD cues and that the stimuli for the EEG recording (which were adapted based on individual participants’ matching performance) were comparable across the two groups.

EEG task
Figure 2b shows performance on the behavioural task completed during the EEG recording. A two-way ANOVA showed that performance was superior for IAD compared to ITD cues, F(1,28) = 18.57, p < .001, ges = 0.062. However, there was no effect of Group, F(1,28) = 3.08, p = .090, ges = 0.090 and no interaction, F(1,28) = 0.00, p = .988, ges = 0.000.
Event related potentials
Figure 3 shows the event related waveforms for the TD and ASD groups for the ITD and IAD stimuli. As expected, auditory stimuli elicited a P1-N1-P2 complex. Differences in the response to Pitch and Control stimuli were evident between 196 and 268 ms (ORN) and between 360 and 564 ms (P400).
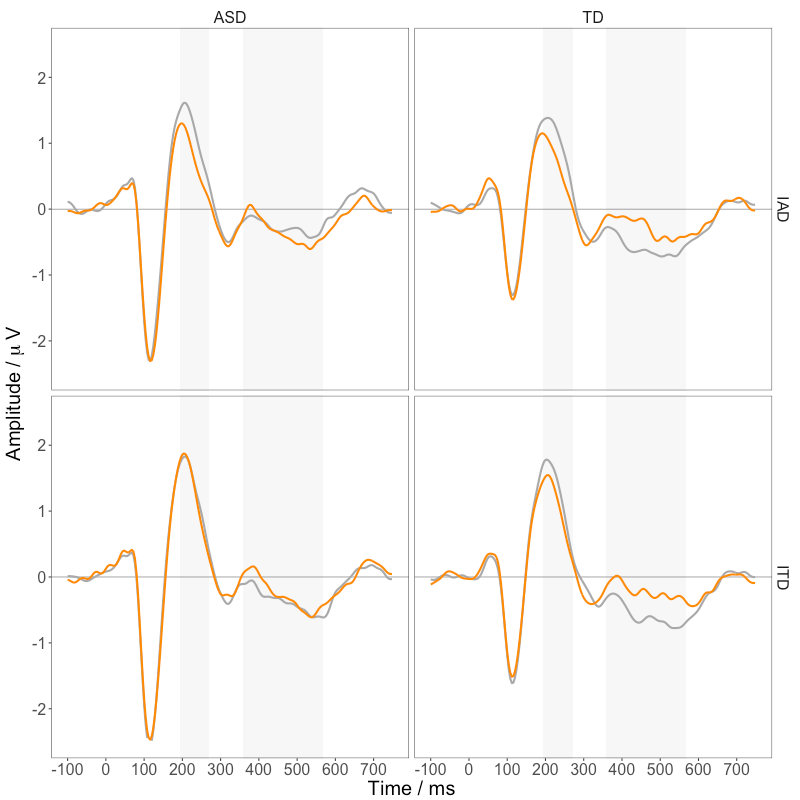
Figure 4 shows the difference waveforms for individual participants in the two groups.
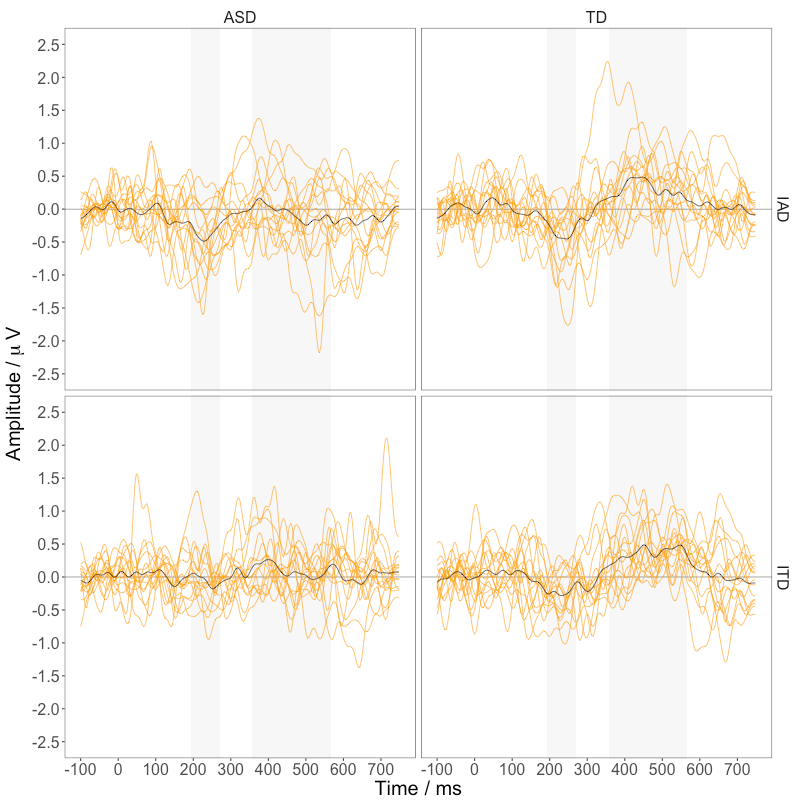
Figure 5 shows the topographies of the ORN and P400 within these two time windows.
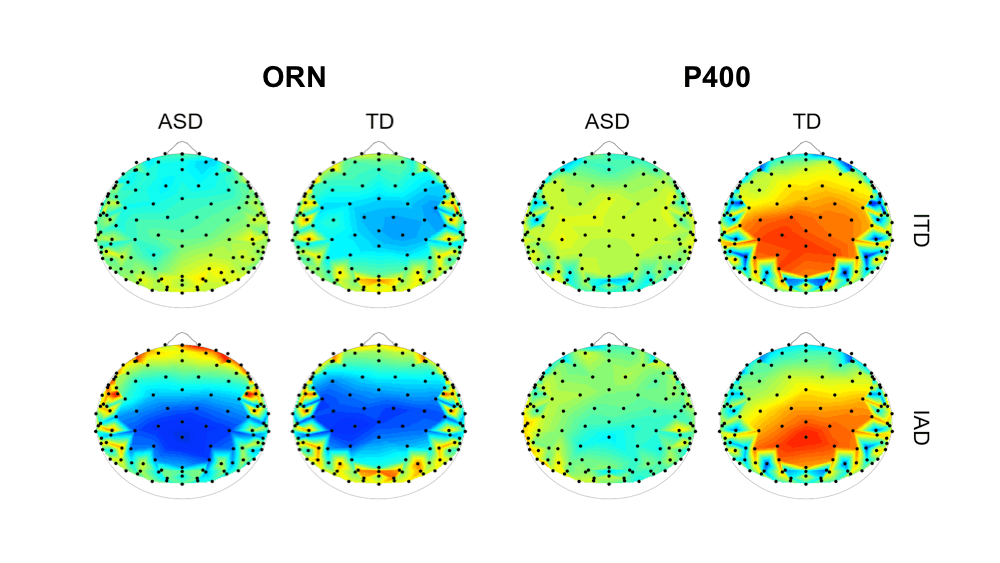
Figure 6 (left panel) shows the ORN magnitude across group and cue type. The TD group produced a significant ORN to both ITD and IAD cues. In contrast, the ASD group only showed an ORN to IAD cues. However, this qualitative pattern was not fully supported by the two-way ANOVA directly comparing the two groups. The ORN was larger for IAD than for ITD stimuli, F(1,28) = 5.97, p = .021, ges = 0.084, but the main effect of Group, F(1,28) = 1.42, p = .243, ges = 0.028, and the interaction between Group and Cue, F(1,28) = 0.10, p = .754, ges = 0.002, were not significant.
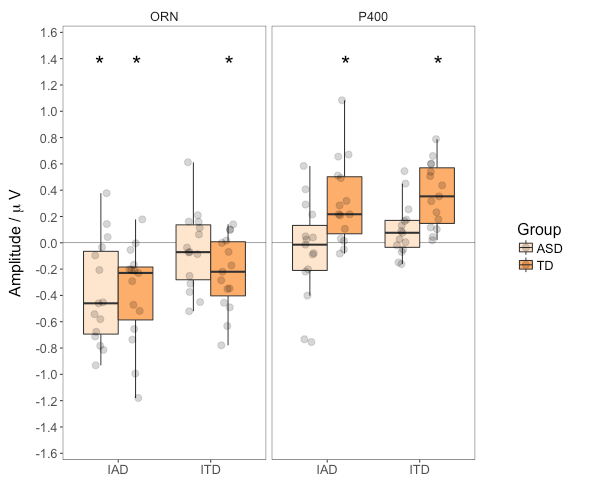
Figure 6 (right panel) shows the P400 magnitude. Participants in the TD group produced a P400 to both cues, whereas those in the ASD group failed to show a P400 to either stimulus. Consistent with these observations, two-way ANOVA showed a significant effect of Group, F(1,28) = 9.03, p = .006, ges = 0.175, but no effect of Cue, F(1,28) = 2.85, p = .102, ges = 0.033, and no interaction, F(1,28) = 0.05, p = .829, ges = 0.001.
Finally, we determined whether performance on the behavioural tasks conducted during EEG recording was correlated with the corresponding ORN or P400. We did this separately for the two cues (ITD and IAD) and groups. None of the correlations were statistically significant, even when uncorrected for multiple comparisons.
Discussion
The human auditory system makes use of both amplitude and timing cues in order to differentiate between sounds emanating from different spatial locations. Our previous EEG and MEG studies (Brock et al., 2013; Lodhia et al., 2014) provided evidence that autistic children and adults have difficulty forming auditory objects on the basis of timing cues. The results from the ITD condition of the current study were broadly consistent with these findings. TD adults showed a significant ORN but adults in the ASD group did not. In contrast, both groups showed a clear ORN to amplitude cues (IAD condition). As noted above, the ORN is considered a measure of auditory object formation, arising when participants are able to infer multiple sources of the incoming sound. The current results indicate, therefore, that autistic adults are able to form distinct auditory objects based on IAD cues even if they do not do so on the basis of ITD cues. In other words, there is no generalised impairment in auditory object formation.
Before proceeding further, it is important to note that the ANOVA directly contrasting cues and groups did not reveal a significant interaction. That is, we cannot say that the ASD group were insensitive to ITD cues relative to IAD cues. Nonetheless, we have now reported three studies in three independent samples in which the TD group have shown robust differential responses to ITD stimuli that was absent in the corresponding ASD group. It is, therefore, worth considering why autistic individuals might experience difficulties forming auditory objects based on ITD cues but not IAD cues.
One possibility is that the neural mechanism involved in computing ITDs (but not IADs) is affected in autism. ITDs are computed in the medial superior olive of the brainstem by means of neural circuits involving delay lines and coincidence detectors and is, therefore, dependent on the precise timing of neural signals. IADs are computed in the lateral superior olive by effectively subtracting the signal from the ipsi- and contralateral ears. A specific disruption of the medial superior olive – or a more general disruption of neural timing – would both affect ITDs more than IADs. However, the good performance of autistic adults on the behavioural tasks indicates that they were at least able to detect the ITDs. It follows that any difficulties processing ITD-related spatial information occurs at a later stage of processing, when the output of the medial superior olive is used to form auditory objects.
In addition to the ORN, we also considered the subsequent P400 response. Participants in the TD group showed – as expected – a significant P400 to both ITD and IAD cues. In contrast, the ASD group failed to show a P400 to either cue. The absence of a P400 response in the ASD group is particularly intriguing in the IAD condition where it followed an intact ORN. The P400 is typically observed only when participants are required to respond to the stimuli and is, therefore, thought to reflect the conscious decision-making process of determining the number of auditory objects (Alain, 2007; Toth et al., 2016). The absence of the P400 in the ASD group, despite adequate behavioural task performance suggests either that this characterisation of the P400 is incorrect, or that the ASD group were performing the task without actually considering the number of auditory objects.
In summary, the current study provides the first clear evidence that autistic individuals are able to use IAD cues to auditory object formation and, as such, do not experience a general inability to form auditory objects. They may, however, experience more specific difficulties using ITD cues and, regardless of the cue, may not engage the later, attention-demanding aspects of auditory object formation. Such difficulties could explain the challenges that many autistic individuals face when confronted with complex, real-world listening environments with implications in particular for spoken language comprehension and its development. Further research with larger sample sizes and perhaps more trials per subject is required to show definitively whether autistic individuals are able to form auditory objects based on ITD cues and, if not, at what stage in processing the breakdown occurs. Given the heterogeneity within the autism population, there may be some variation across individuals and so future research could also consider sensory and cognitive correlates of auditory object formation as well as more detailed and rigorous clinical assessment of participants. Nonetheless, the current study provides a further illustration of the potential of the auditory scene analysis framework for understanding the atypical auditory experiences of individuals on the autism spectrum.
References
Alain, C. (2007). Breaking the wave: effects of attention and learning on concurrent sound perception. Hear. Res., 229(1), 225-236.
Alain, C., Arnott, S.R., & Picton, T.W. (2001).Bottom-up and top-down influences on the auditory scene analysis: evidence from event related brain potentials. J. Exp. Psychol., 27(5), 1072-1089.
Alarcon, G., Guy, C.N., Binnie, C.D. (2000). A simple algorithm for a digital three-pole Butterworth filter of arbitrary cut-off frequency: application to digital electroencephalography. J. Neuro Met., 104(1), 35–44.
Alcantara, J.I., Cope, T.E., Cope, W., & Weisblatt, E. J. (2012). Auditory temporal- envelope processing in high-functioning children with Autism Spectrum Disorder. Neuropsychologia, 50, 1235-1251.
Alcantara, J.I., Weisblatt, E.J.L., Moore, B.C.J., & Bolton, P.F. (2004). Speech in noise perception in high functioning individuals with autism or Asperger’s syndrome. J. Child Psycholo. Psychiatry., 45(6), 1107-1114.
American Psychiatric Association. (1994). Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV). Washington, DC: American Psychiatric Publishing.
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). Washington, DC: Author.
Berument, S. K., Rutter, M., Lord, C., Pickles, A., & Bailey, A. (1999). Autism screening questionnaire: diagnostic validity. British. J. Psychiatry, 175(5), 444-451.
Bilsen, F. A. (1976). Pronounced binaural pitch phenomenon. J. Acoust. Soc. Am., 59, 467-468.
Boatman, D., Alidoost, M., Gordan, B., Lipsky, F., & Zimmerman, W. (2001). Enhanced pitch sensitivity in individuals with autism: a signal detection analysis. J. Cogn. Neurosci, 15:226–235.
Bregman, A.S. (1990). Auditory scene analysis. The perceptual organization of sound. Cambridge, Massachusetts: The MIT Press.
Brock, J., Bzishvili, S., Reid, M., Hautus, M.J., & Johnson, B.W. (2013). Brief Report: Atypical Neuromagnetic Responses to Illusory Auditory Pitch in Children with Autism Spectrum Disorders. J. Autism Dev. Disord., DOI 10.1007/s10803-013-1805-z.
Clapp, W.C., Johnson, B.W., & Hautus, M. J. (2007). Graded cue information in dichotic pitch: effects on event-related potentials. NeuroReport, 18 (4), 365-638.
Cramer, E.M., & Huggins, W.H. (1958). Creation of pitch through binaural interaction. J. Acoust. Soc. Am., 30(5), 413-417.
Dougherty, R., Cynader, MS., Bjornson, B. H. Edgell, D., & Giaschi, D. E. (1998). Dichotic pitch: a new stimulus distinguishes normal and dyslexic auditory function. NeuroReport, 9(13), 3001-3005.
Grandin, T. & Scarinano, M.M., (1986). Emergence: Labelled autistic. New York: Arena Press.
Groen, W.B., van Orsouw, L., Huurne, N., Swinkels, S., van der Gaag, R.J., Buitelaar, J.K & Zwiers, M.P. (2009). Intact spectral but abnormal temporal processing of auditory stimuli in autism. J. Autism Dev. Disord., 39 (3), 742-750.
Foxton, J. M., Stewart, M.E., Barnard, L., Rodgers, J., Young, A.H., O’Brien, Griffiths, T.D. (2003). Absence of auditory ‘global interference’ in autism. Brain, 126, 2703-2709.
Hautus, M. J., Johnson, B.W., & Colling, L.J. (2009). Event-related potentials for interaural timing differences and spectral cues. NeuroReport, 20, 951-956.
Hautus, M.J, Johnson. B.W. (2005). Object-related brain potentials associated with the perceptual segregation of dichotically embedded pitch. J. Acoust. Soc. Am., 117(1), 275–280. DOI 10.1121/1.1828499.
Jervis, B.W., Nichols, M.R., Allen, E.M., Hudson, N.R., & Johnson, T. E. (1985). The assessment of two methods for removing eye movement artifacts from the EEG. Electro. Clin. Neurophysiol., 61, 444-452.
Johnson, B.W., & Hautus, M. J. (2010). Processing of binaural spatial information in human auditory cortex: neuromagnetic responses to interaural timing and level difference. Neuropsychologia, 48, 2610-2619.
Johnson, B.W., Hautus, M., & Clapp, WC. (2003). Neural activity associated with binaural processes for the perceptual segregation of pitch. Clin. Neurophysiol, 114, 2245-2250.
Johnson, B.W., Hautus, M.J., Duff, D.J., & Clapp, W.C. (2007). Sequential processing of interaural timing differences for sound source segregation and spatial localization: Evidence from cortical event-related potentials. Psychophysiology, 44, 541-551.
Kujala, T., Kuuluvainen, S., Saalasti, S., Jansson-Verkasalo, E., von Wendt, L., & Lepistö, T. (2010). Speech-feature discrimination in children with Asperger syndrome as determined with the multi-feature mismatch negativity paradigm. Clin.Neurophysiol., 121, 1410-1419.
Kulesza, R.J. & Mangunay, K. (2008). Morphological features of the medial superior olive in autism. Brain Res., 1200, 132-137.
Landon, J., Shepherd, D., & Lodhia, V. (2016). A qualitative study of noise sensitivity in adults with autism spectrum disorder. Res. Autism Spec. Dis., 32, 43-52.
Lin, F.I., Shirma, A., Kato, N., & Kashino, M. (2016). The singular nature of auditory and visual scene analysis in autism. Phil. Trans. R. Soc. B., 372: 20160115. http://dx.dio.org/10.1098/rstb.2016.0115
Lodhia, V., Brock, J., Johnson, B. W., & Hautus, M. J. (2014). Reduced object related negativity response indicates impaired auditory scene analysis in adults with autistic spectrum disorder. PeerJ 2:e261; DOI 10.7717/peerj.261
Macmillan NA, Creelman CD. 2005. Detection theory: a user’s guide, 2nd edn. Mahwah, New Jersey: Lawrence Erlbaum Associates.
Oldfield, R.C. (1971). The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia, 9, 97–113 DOI 10.1016/0028-3932(71)90067-4.
Pratt, H, Polyakov, A., & Kontorovich, L. (1997). Evidence for separate processing human brainstem of interaural intensity and temporal disparities for sound lateralizations. Hear. Res., 108, 1-8.
Rogers, S.J., & Ozonoff, S. (2005). Annotation: What do we know about sensory dysfunction in autism? A critical review of the empirical evidence. J. Child Psychol. Psychiatry, 46, 1255-1268. DOI: 10.1007/s10803-012-1608-7.
Rodier, P.M., Ingram, J.L., Tisdale, B., Nelson, S., & Romano, J. (1996). Embryological origin for autism: developmental anomalies of the cranial nerve motor nuclei. J. Compara. Neurolo., 370 (2), 247-261.
Rosenhall, U., Nordin, V., Sandstrom, M., Ahlsen, G., & Gillberg, C. (1999). Autism and hearing loss. J. Autism Dev. Dis., 29, 349-357.
Smith, P.H., Joris, P.X., & Yin, T.C. (1993). Projections of physiologically characterized spherical bushy cell axons from the cochlear nucleus of the cat: evidence for delay lines to the medial superior olive. J. Compara. Neurolo., 33, 245-260.
Talay-Ongan A, Wood, K. (2000). Unusual sensory sensitivities in autism: a possible crossroads. Intern. J. Disability, Dev. Edu., 47(2), 201–212 DOI 10.1080/713671112.
Teder-Salejarvi, W.A., K.L. Pierce, E. Courchesne and S.A. Hillyard. (2005). Auditory spatial localization and attention deficits in autistic adults. Cog. Brain Res., 23, 221-234.
Tóth, B., Kocsis, Z., Háden, G. P., Szerafin, Á., Shinn-Cunningham, B. G., & Winkler, I. (2016). EEG signatures accompanying auditory figure-ground segregation. Neuroimage, 141, 108-119.
Tucker, D.M. (1993). Spatial sampling of head electrical fields: the geodesic sensor net. Electroencephalo. & Clin. Neurophysiol., 87(3), 154–163 DOI 10.1016/0013-4694(93)90121-B.
Wechsler, D. (1999). Wechsler abbreviated scale of intelligence (WASI). New York: The Psychological Corporation.
Yin, T.C.T & Kuwada, S. (1984). Neuronal mechanisms of binaural interaction. In G.M. Edelman (Ed.), Dynamic aspects of neocortical function (pp. 263-313). New York: Wiley.
Acknowledgements
The authors gratefully acknowledge the assistance and support from Autism NZ, Altogether Autism, Autism House, the participants and their families for supporting this research. The work of BWJ and JB was supported by the Australian Research Council (ARC) Centre of Excellence for Cognition and its Disorders (CE110001021). JB was supported by an ARC Australian Research Fellowship (DP098466). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
