Brock, J., Bzishvili, S., Reid, M., Hautus, M., & Johnson, B. W. (2013). Atypical neuromagnetic responses to illusory auditory pitch in children with autism spectrum disorder. Journal of Autism and Developmental Disorders, 43, 2726-2731. Preprint PDF
Atypical auditory perception is a widely recognised but poorly understood feature of autism. In the current study, we used magnetoencephalography to measure the brain responses of 10 autistic children as they listened passively to dichotic pitch stimuli, in which an illusory tone is generated by sub-millisecond inter-aural timing differences in white noise. Relative to control stimuli that contain no inter-aural timing differences, dichotic pitch stimuli typically elicit an Object Related Negativity (ORN) response, associated with the perceptual segregation of the tone and the carrier noise into distinct auditory objects. Autistic children failed to demonstrate an ORN, suggesting a failure of segregation; however, comparison with the ORNs of age-matched typically developing controls narrowly failed to attain significance. More striking, the autistic children demonstrated a significant differential response to the pitch stimulus, peaking at around 50 milliseconds. This was not present in the control group, nor has it been found in other groups tested using similar stimuli. This response may be a neural signature of atypical processing of pitch in at least some autistic individuals.
Autistic individuals often report atypical auditory perceptual experiences including acute sensitivity or emotional responses to certain sounds, as well as difficulty making sense of complex acoustic environments (e.g., Alcántara, Weisblatt, Moore, & Bolton, 2004; Cesaroni & Garber, 1999). These first hand accounts are corroborated by parental reports (e.g., Wiggin, Robins, Bakeman, Adamson, 2009), clinical observations (e.g., Greenspan & Weider, 1997), and video analysis (Baranek, 1999) indicating both hyper- and hyporesponsiveness to auditory stimuli. Experimental studies have revealed lower discomfort thresholds for pure tones (Khalfa et al 2004), enhanced performance on pitch discrimination and memory tasks (e.g., Bonnel et al., 2010), and difficulties processing speech in noise (Alcántara et al., 2004).
Numerous studies have investigated auditory brain responses in autism using event-related potentials (ERPs), although the pattern of results across studies is highly inconsistent (Haesen, Boets, & Wagemans, 2011). For example, a number of studies have reported that autistic individuals have atypically large mismatch negativity (MMN) responses to deviants tones in a stream of otherwise identical tones (e.g., Ferri et al., 2003; Lepistö et al, 2005); but other studies have failed to replicate this finding (e.g., Cepioniene et al., 2003; Jansson-Verkasalo et al., 2003). Of particular relevance to the current study, Lepistö et al. (2009) reported that autistic and non-autistic adults showed similar MMN responses to deviants in a sequence of tones. However, only the control participants showed an MMN when the same stimuli were played against a background of higher tones. This, the authors suggested, indicated a failure of autistic adults to segregate the tones into separate auditory streams (cf. Bregman, 1990).
The initial aim of the current study was to further investigate auditory segregation in autism. We used magnetoencephalography (MEG) to investigate the brain responses of autistic children to dichotic pitch stimuli – white noise bursts in which a narrow frequency band is shifted in time by a fraction of a millisecond in one ear only (Dougherty, Cynader, Bjornson, Edgell, & Giaschi, 1998). This inter-aural timing difference typically results in the illusory perception of a pitch sound in the leading ear, spatially segregated from the “carrier” noise burst. Previous ERP and MEG studies using similar stimuli have identified a response at around 150-300 milliseconds after sound onset that occurs independent of attention to the stimulus and differentiates between dichotic pitch stimuli and control stimuli with no inter-aural timing difference (Hautus & Johnson, 2005; Johnson & Hautus, 2010). This response is assumed to be equivalent to the Object Related Negativity (ORN) elicited by a mistuned harmonic sound (Alain, 2007) and, as such, is thought to reflect the cortical processes associated with automatic segregation of the ‘auditory scene’ into separate objects (Johnson, 2011).
Given the well-documented auditory processing difficulties experienced by autistic individuals, and the suggestive evidence for deficits in auditory segregation, we predicted that children with an autism diagnosis would demonstrate a reduced ORN response relative to age-matched, typically developing control participants.
Method
Subjects
10 autistic children (7 boys) aged between 8 and 11 years (mean = 115 months, SD = 13) were recruited from Autism Spectrum Australia and the Macquarie University Special Education Centre. All 10 were attending special education schools or “satellite classes” and had received DSM-IV diagnoses of autistic disorder, Asperger’s disorder, or pervasive developmental disorder – not otherwise specified from experienced psychologists or psychiatrists. In nine out of ten cases, the autism spectrum diagnosis was corroborated by an above-threshold score of 15 or greater on the Social Communication Questionnaire (Rutter, Bailey, & Lord, 2003). The tenth child, a member of a multiplex family, had a score of 12, just below the cut-off. Exclusion of this child had no effect on the significance or otherwise of group comparisons. Indeed, the group difference we report remained significant even if we excluded the four children with SCQ scores below 22. Autistic children had mean standard scores of 92.7 (SD = 20.7) on the Peabody Picture Vocabulary Test – 2nd edition; 94.1 (23.9) on the Test for Reception of Grammar – 2nd edition (reference means 100, SD = 15). They also had standard scores of 5.4 (4.4) on the Sentence Repetition subtest of the Clinical Evaluation of Language Fundamentals – 4th edition; and 9.1 (3.2) on the Matrices subtest of the Wechsler Intelligence Scale for Children – 4th edition (reference means 10, SD = 3).
Brain responses of the autistic children were compared with those of 10 typically developing children (mean age = 117 months, SD = 14) drawn from a larger study using the same stimuli and MEG system that is reported elsewhere (Johnson et al., 2013). Given that the paradigm was entirely passive, requiring no performance element or attention to the stimuli, we made no attempt to match groups on measures of verbal or nonverbal ability. There is, however, considerable evidence for developmental changes in auditory evoked responses in this age range (e.g., Ponton et al., 2000). We therefore took special care selecting control participants to match the autistic children individually on chronological age (p = 0.745, Cohen’s d = 0.157, variance ratio = 1.16) as well as sex.
For both groups, auditory thresholds were checked using an Otovation Amplitude T3 series audiometer (Otovation LLC, King of Prussia, PA). All participants met our cut-off of a pure-tone average threshold less than 15 dB HL.
Stimuli
Stimuli were 500 ms white noise bursts, filtered between 400 and 800 Hz. For the dichotic pitch stimulus, an interaural timing difference was introduced such that a narrow band of the noise burst, centred on 600 Hz, was delayed in the left channel. This typically results in perception of a 600 Hz tone in the leading (right) ear. For the control stimulus, there was no inter-aural timing difference and so no tone could be perceived. Full details of stimulus generation are provided elsewhere (Johnson et al., 2013). Stimuli were delivered to listeners using insert earphones (Model ER-30, Etymotic Research Inc., Elk Grove Village, IL). The level of the sounds was adjusted using programmable attenuators (Model PA4, Tucker Davis Technologies, Alachua, FL) to yield 70 dB SPL at the eardrum.
MEG acquisition
Prior to acquisition, MEG marker coils were placed on the participant’s head and marker coil positions and head shape were measured with a pen digitizer (Polhemus Fastrack, Colchester, VT). For typically developing children, marker coils were affixed to an MEG-compatible EEG cap (BrainProducts GmbH, Gilching, Germany). This is non-ferrous and so cannot interfere with recordings of brain responses. For autistic participants, the coils were affixed to an elastic cap (essentially a fabric swimming cap). All measurements were carried out with the subject in a supine position in a magnetically shielded room (Fujihara Co. Ltd., Tokyo, Japan) using the KIT-Macquarie MEG160 (Model PQ1160R-N2, KIT, Kanazawa, Japan), which consisted of 160 coaxial first-order gradiometers with a 50 mm baseline (Kado et al., 1999). MEG data were acquired with a sampling rate of 1000 Hz and filter bandpass of 0.03-200 Hz.
Procedure
During the MEG recordings, children were instructed to ignore the experimental stimuli. To maintain compliance with the recording procedure, they watched a movie of their choice, played with low-level video sound. Four 10-minute blocks of randomly interleaved control and pitch stimuli were presented with a jittered interstimulus interval of between 800 and 1200 ms. Each block contained 216 stimuli (113 of each type), for a total of 864 trials. Stimulus blocks were presented consecutively with a short interval in between during which head position was measured. The head movement tolerance threshold was <5 mm from start to end of a block.
Analysis
MEG data were processed and analysed off-line using BESA Research version 5.3.1 (MEGIS Software GMbH, Grafelfing, Germany). Data were segmented and averaged into 600 ms epochs including a pre stimulus baseline of 100 ms. Averaged MEG data (event-related magnetic fields) were filtered with a bandpass of 0.16 – 40 Hz. Artefacts, including blinks and eye-movements, were rejected using the artefact scan tool in BESA 5.2.4, which rejects trials based on abnormally high amplitudes or abrupt rises or falls in amplitude (gradients). For each subject and condition, at least 90% of trials survived artefact rejection.
Epoched MEG data were projected onto two bilateral sources with approximate Talairach coordinates (based on a template brain) of +/- 47mm, -24.4 mm, 14.9 mm (X, Y, Z), corresponding to the transverse temporal gyri (BA41, primary auditory cortex) (see Johnson et al., 2013). Although source locations were fixed across individuals, source orientations were derived from each individual’s own data by adjusting the orientation of the first tangential component to the maximum amplitude within a window of 80-105 ms, the latency of the rising edge of the M100 component for each subject. This approach rests on the assumption that individual variation in source location is relatively small and has a minimal effect on recordings in comparison to the much larger variation in source orientation due to differences in gyral anatomy and functional representation (Scherg and von Cramon, 1986). Source waveforms were analysed using the BESA source waveform toolbox for MATLAB (MEGIS Software GMbH, Grafelfing, Germany). For each difference waveform, 95% confidence intervals were determined using the bootstrap bias-corrected and adjusted (BCa) method (Efron & Tibshirani, 1993) with 1000 bootstrap samples.
Results
Figure 1 shows the left and right hemisphere waveforms for each group as a function of condition. Figure 2 shows the difference waveform (Control – Pitch) for each group and each hemisphere, together with 95% confidence intervals. Consistent with predictions, children in the ASD group failed to show a differential response in either hemisphere during the ORN window, which we took to be any time after 150 ms. In contrast, the typically developing children did show an ORN effect in the right hemisphere only1. However, this was only marginally significant and, when difference waveforms for the two groups were compared directly, the group differences were not significant, with 95% confidence intervals (as shown in the lower panel of Figure 2) including zero throughout the ORN window. We re-analysed the data including all 16 typically developing children. Although there was now a much clearer ORN effect in the typically developing group, the group differences in the ORN window remained non-significant.
 Figure 1: Evoked responses to Pitch (black) and Control (grey) stimuli in the left and right hemispheres
Figure 1: Evoked responses to Pitch (black) and Control (grey) stimuli in the left and right hemispheres
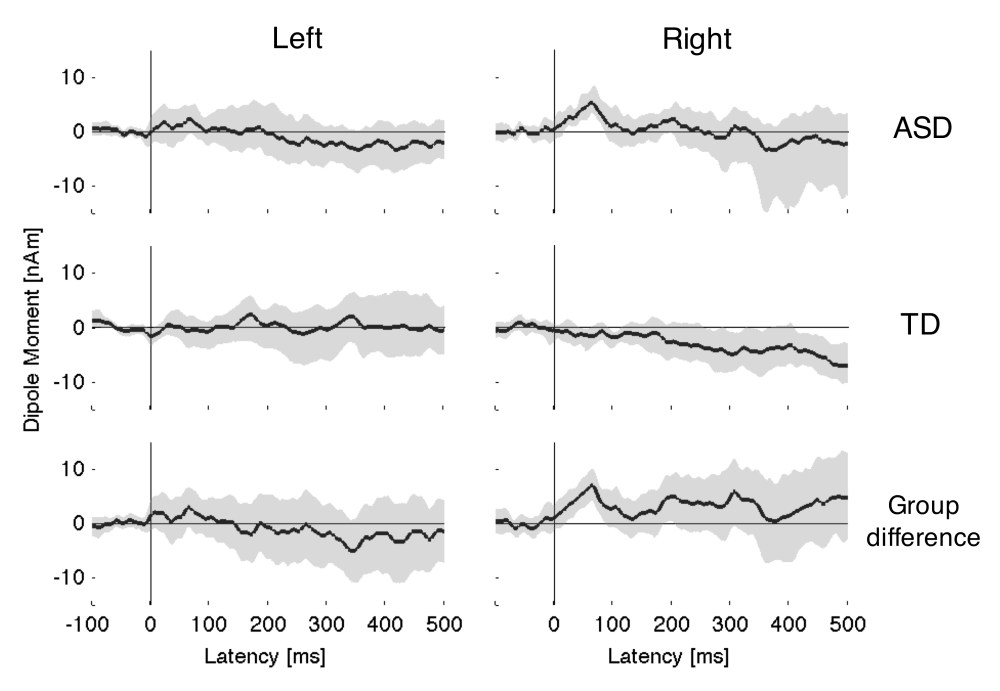
Figure 2: Difference waveforms (Control minus Pitch) for left and right hemispheres with 95% confidence intervals (shaded regions)
Unexpectedly, the ASD group showed an early difference in their responses to the pitch and control stimuli in the right hemisphere, peaking at 63 milliseconds. Allowing for the fixed 13 ms conduction delay in our sound delivery system, equates to a response at 50 milliseconds post-onset. This component was absent from the control group. Group differences were significant and remained so even when a more stringent 97.5% confidence interval was applied to correct for multiple comparisons. Further, this group effect remained significant when we systematically omitted each autistic participant, confirming that it was not driven by any single outlier.
Discussion
Motivated by converging evidence for atypical auditory processing in autism, the current study used MEG to measure the brain responses of autistic children to illusory pitch stimuli. Our initial prediction was that children with autism would evidence a reduction in the magnetic counterpart of the ORN, indicating a failure to segregate the “auditory scene” into distinct objects. Consistent with this, we found no evidence of an ORN in the difference waveforms of autistic children. However, comparison with age-matched typically developing children narrowly failed to show a significant group effect on the magnitude of the ORN. Given the relatively small sample size, this may reflect a lack of statistical power. We cannot, therefore, draw any firm conclusions about the reduction or otherwise of the ORN in autism based on the current data, although the trend is consistent with previous reports of reduced auditory segregation in autistic adults (Lepistö et al., 2009).
The more compelling findings came, unexpectedly, from an earlier time window. Despite failing to show an ORN response, the autistic children did show a significant differential response to the pitch versus control stimuli, peaking at around 50 milliseconds post-stimulus onset in the right hemisphere only. It is not clear at this point why the response should be right-lateralized or whether this would reverse if the pitch was perceived to the left of space. However, we note that the finding is consistent with the notion of right hemisphere specialization for pitch processing (e.g., Zatorre & Gandour, 2008) and the right-lateralized ORN in the typically developing control group. Critically, the early response was not found in typically developing controls in either hemisphere. Nor was it found in dyslexic children (Johnson et al., 2013) or healthy, non-autistic adults (Johnson & Hautus, 2010) tested in other studies using the same MEG system and the same or similar stimuli.
Potential concerns here are that the ASD data may have been somewhat noisier than for other groups, that the dipoles in our source model may have been placed suboptimally for children in the ASD group, or that the group differences are somehow a reflection of differences in the child’s position in the MEG dewar (helmet). However, in order to explain the presence of the earlier component in the difference waveform of the ASD group, such artefacts would have to systematically affect one condition more than the other. Considering that the pitch and control stimuli were randomly interleaved, this seems unlikely. Our provisional interpretation, therefore, is that the early dichotic response in our ASD group may indicate atypical early stages of auditory cortical processing in at least a subgroup of autistic individuals.
A number of previous studies of autism have considered auditory evoked responses in a similar latency range, specifically the P1 and M50 measured by EEG and MEG respectively (e.g., Ceponiene et al., 2003; Jansson-Verkasalo et al., 2003; Lepistö et al., 2005, 2009; Oram-Cardy, Ferrari, Flagg, Roberts, & Roberts, 2004; Roberts et al., 2010). In apparent contradiction of the current results, these indicate either age-appropriate or diminished responses in autistic individuals. However, our dichotic pitch stimuli are quite different to the stimuli (generally pure tones) used in these previous studies. Most importantly, perhaps, our analyses focused not on the response to a single stimulus but on the difference between pitch and control stimuli. Because these had identical spectral profiles and amplitude envelopes, the difference waveform represents the response to the (illusory) pitch, independent of the brain response to the onset of acoustic energy. Thus, the current findings speak specifically to the early stages of pitch encoding rather than the response to sound onset as measured in previous studies.
The study is limited by the relatively modest size of the ASD group. This is of particular relevance to the ORN analysis, where group differences may become significant with larger sample sizes. Small sample sizes also increase the likelihood of false positive results, although we have shown that the central finding of an atypical early response to the pitch stimulus is not driven by any single individual in the ASD group. As discussed above, we have also ruled out a number of potentially confounding factors that could explain this group difference. Nonetheless, given the unanticipated nature of this finding, it should be treated with caution – at least until it is replicated. It is also unclear at this stage how specific the atypical early response is to autism and how consistent it is across different individuals within the autism population. Nor is its functional significance clear. Although it is tempting to assume that enhanced cortical sensitivity to illusory pitch in autism is linked to atypical auditory perception, we have not yet linked the two at the level of individuals. These and other issues we are addressing in ongoing research.
References
Alain, C. (2007). Breaking the wave: effects of attention and learning on concurrent sound perception. Hearing Research, 229(1-2), 225-236.
Alcántara, J. I., Weisblatt, E. J. L., Moore, B. C. J., & Bolton, P. F. (2004). Speech-in-noise perception in high-functioning individuals with autism or Asperger’s syndrome. Journal of Child Psychology and Psychiatry, 45(6), 1107–1114.
Baranek, G. (1999). Autism during infancy: A retrospective video analysis of sensory-motor and social behaviors at 9–12 months of age. Journal of Autism and Developmental Disorders, 29, 213–224.
Bonnel, A., McAdams, S., Smith, B., Berthiaume, C., Bertone, A., Ciocca, V., Burack, J. A., & Mottron, L. (2010). Enhanced pure-tone pitch discrimination among persons with autism but not Asperger syndrome. Neuropsychologia, 48, 2465–2475.
Bregman, A.S. (1990). Auditory scene analysis. The perceptual organization of sound. Cambridge, Massachusetts: The MIT Press.
Ceponiene, R., Lepistö, T., Shestakova, A., Vanhala, R., Alku, P., Naatanen, R., & Yaguchi, K. (2003). Speech-sound-selective auditory impairment in children with autism: They can perceive but do not attend. Proceedings of the National Academy of Sciences of the United States of America, 100, 5567–5572.
Cesaroni, L. & Garber, M. (1991). Exploring the experience of autism through firsthand accounts. Journal of Autism and Developmental Disorders, 21,303-313.
Dougherty, R. F., Cynader, M. S., Bjornson, B. H., Edgell, D., & Giaschi, D. E. (1998). Dichotic pitch: a new stimulus distinguishes normal and dyslexic auditory function. Neuroreport, 9(13), 3001-3005.
Efron, B. & Tibshirani, R. J. (1993) An introduction to the bootstrap. New York: Chapman & Hall.
Ferri, R., Elia, M., Agarwal, N., Lanuzza, B., Musumeci, S. A., & Pennisi, G. (2003). The mismatch negativity and the P3a components of the auditory event-related potentials in autistic low-functioning subjects. Clinical Neurophysiology, 114, 1671–1680.
Greenspan, S., & Weider, S. (1997). Developmental patterns and out- comes in infants and children with disorders in relating and communicating. A chart review of 200 cases of children with autistic spectrum diagnoses. Journal of Developmental and Learning Disorders, 1, 87–141.
Haesen, B., Boets, B., & Wagemans, J. (2011). A review of behavioural and electrophysiological studies on auditory processing and speech perception in autism spectrum disorders. Research in Autism Spectrum Disorders, 5, 701–714.
Hautus, M. J., & Johnson, B. W. (2005). Object-related brain potentials associated with the perceptual segregation of a dichotically embedded pitch. Journal of the Acoustical Society of America, 117, 275-280.
Jansson-Verkasalo, E., Ceponiene, R., Kielinen, M., Suominen, K., Jantti, V., Linna, S. L., Moilanen, I., & Näätänen, R. (2003). Deficient auditory processing in children with Asperger syndrome, as indexed by event-related potentials. Neuroscience Letters, 338, 197–200.
Johnson. B.W. (2011). Processing of Binaural Information in Human Auditory Cortex, Advances in Sound Localization, P. Strumillo (Ed.), ISBN: 978-953-307-224-1, InTech, Available from: http://www.intechopen.com/articles/show/title/processing-of-binaural-information-in-human-auditory-cortex.
Johnson, B. W., & Hautus, M. J. (2010). Processing of binaural spatial information in human auditory cortex: neuromagnetic responses to interaural timing and level differences. Neuropsychologia, 48(9), 2610-2619.
Johnson, B. W., McArthur, G., Hautus, M., Reid, M., Brock, J., Castles, A., & Crain, S. (2013). Lateralized auditory brain function in children with normal reading ability and children with dyslexia. Neuropsychologia, 51, 633-641.
Kado, H., Higuchi, M., Shimogawara, M., Haruta, Y., Adachi, Y., Kawai, J., Ogata, H., & Uehara, G. (1999). Magnetoencephalogram systems developed at KIT. IEEE Transactions on Applied Superconductivity, 9(2), 4057-4062.
Khalfa, S., Bruneau, N., Roge, B., Georgieff, N., Veulliet, E., Adrien, J.-L., Barthelemy, C., & Collet, L. (2004). Increased perception of loudness in autism. Hearing Research, 198, 87–92.
Lepistö, T., Kuitunen, A., Sussman, E., Saalasti, S., Jansson-Verkasalo, E., Nieminen-von Wendt, T., & Kujala, T. (2009). Auditory stream segregation in children with Asperger syndrome. Biological Psychology, 82, 301-307.
Lepistö,T., Kujala, T., Vanhala, R., Alku, P., Huotilainen, M., & Naatanen, R., (2005). The discrimination of and orienting to speech and non-speech sounds in children with autism. Brain Research 1066, 147–157.
Oram Cardy, J.E., Ferrari, P., Flagg, E.J., Roberts, W., & Roberts, T.P. (2004). Prominence of M50 auditory evoked response over M100 in childhood and autism. Neuroreport, 15, 1867–1870.
Ponton, C. W., Eggermont, J. J., Kwong, B., & Don, M. (2000). Maturation of human central auditory system activity: evidence from multi-channel evoked potentials. Clinical Neurophysiology, 111, 220-236.
R oberts T.P., Khan, S. Y., Rey, M., Monroe, J. F., Cannon, K., Blaskey, L., Woldoff, S., Qasmieh, S., Gandal, M., Schmidt, G. L., Zarnow, D. M., Levy, S. E., & Edgar, J. C. (2010). MEG detection of delayed auditory evoked responses in autism spectrum disorders: Towards an imaging biomarker for autism. Autism Research, 3, 8 –18.
Rutter M, Bailey A, Lord C (2003) SCQ: The Social Communication Questionnaire – Manual. Los Angeles, CA: Western Pscyhological Services.
Scherg, M., & Von Cramon, D. (1986). Evoked dipole source potentials of the human auditory cortex. Electroencephalography and clinical Neurophysiology, 65(5), 344-360.
Wiggin, L. D., Robins, D. L., Bakeman, R., & Adamson, L. B. (2009). Sensory Abnormalities as Distinguishing Symptoms of Autism Spectrum Disorders in Young Children. Journal of Autism and Developmental Disorders, 39, 1087–1091.
Zatorre, R. J. & Gandour, J. T. (2008). Neural specializations for speech and pitch: moving beyond the dichotomies. Philosophical Transactions of the Royal Society, B, Biological Sciences, 363, 1087-1104.
Footnotes
- In an earlier MEG study of dichotic pitch perception in adults, Johnson and Hautus (2010) failed to find evidence of lateralization of the ORN. However, these authors used stimuli in which the noise was lateralized to the opposite side of space to the tone, rather than being perceived centrally as in the current study.
Author Note
We thank the children and their families as well as the staff of Autism Spectrum Australia and Macquarie University Special Education Centre for their participation in the study. The work was supported by an Australian Research Council (ARC) Australian Research Fellowship and Discovery Project (DP098466), an ARC Linkage Infrastructure Equipment and Facilities Grant LEO668421, and the ARC Centre of Excellence in Cognition and its Disorders (CE110001021). The authors gratefully acknowledge the collaboration of Kanazawa Institute of Technology and Yokogawa Electric Corporation in establishing the KIT-Macquarie MEG laboratory.
